Developed as a primary method to share information with federal, state, territorial, and local public health practitioners, clinicians, and public health laboratories, CDC’s Health Alert Network (HAN) is the primary method to share information on urgent public health incidents. The State of West Virginia has a similar system (WV-HAN) designed to share information on urgent public health incidents specific to West Virginia as well.
Below you will find information on the latest Health Alerts, both nationally and specific to West Virginia, as they occur.
Fayette, Kanawha-Charleston Health Departments Share Well Testing Findings From Paint Creek Spill
Environmental health specialists from the Fayette County and Kanawha-Charleston health departments hand delivered and mailed letters this week to Paint Creek-area residents whose water wells were tested for a chemical spilled on the West Virginia Turnpike in late August.

Please Safety Check Your Heart Health Before Going Hunting
Like many West Virginians, I, too, am an avid hunter. I share this information about myself because I understand the lure of the mountains this time of year. When the leaves put on their fall colors, my mind turns to the woods. I want to be nowhere else, and I know my fellow sportsmen and women feel the same.
As a longtime emergency physician, I’ve seen a tragic side of hunting, too. It forces me to have painful conversations with families who have just lost loved ones because they had a heart attack or stroke while hunting and couldn’t get help quickly enough. This little-known consequence of a tradition so many of us love is something we don’t like to think or talk about, but we should.

HEALTH ADVISORY #205 Acute Flaccid Melitis (AFM)
Acute Flaccid Myelitis (AFM) is a reportable condition in West Virginia. Suspected cases should be reported to the local health department (LHD) per the West Virginia Reportable Disease Rule (64 CSR7). Surveillance has shown that AFM cases generally peak in the months of September and October. A biennial pattern has been observed with a larger number of cases reported in 2014, 2016, and 2018. In 2020, cases did not increase likely due to pandemic mitigation measures. Health officials are unsure what to expect in 2022. Public health partners and healthcare providers should be aware of the symptoms of AFM and the related resources to assist with identifying, reporting, and collecting specimens of suspected AFM cases at any time.

HEALTH ALERT #204 CDC Recommends Omicron COVID-19 Booster
On September 1, 2022, the Centers for Disease Control and Prevention (CDC) endorsed the Advisory Committee on Immunization Practices’ (ACIP) recommendations for use of the COVID-19 Omicron updated boosters (i.e., bivalent boosters) from Pfizer-BioNTech for people ages 12 years and older and from Moderna for people ages 18 years and older.

Variant Influenza Virus Infections: Recommendations for Identification, Treatment, and Prevention for Summer and Fall 2022
The Centers for Disease Control and Prevention (CDC) is issuing this Health Alert Network (HAN) Health Advisory to provide updates on recent variant1 influenza virus infections and summarize CDC’s recommendations for identification, treatment, and prevention of variant influenza virus infection for the summer and fall of 2022.

HEALTH ADVISORY #203 Variant Influenza Detected In WV
The 2022 fair season in West Virginia is underway. Past agricultural fairs across the United States have been linked to human infections caused by variant influenza A viruses that are commonly found in swine.

HEALTH ADVISORY #202 WV Monkeypox Outbreak Update
Since late May 2022 the Centers for Disease Control and Prevention (CDC) has been tracking cases of monkeypox that have been reported in the U.S. and other countries without a history of monkeypox activity.
HEALTH ADVISORY #201 CDC Supports Authorization of Novavax New COVID-19 Vaccine for Adults
On July 19, 2022, the Centers for Disease Control and Prevention (CDC) endorsed the Advisory Committee on Immunization Practices (ACIP) recommendation that the Novavax COVID-19 vaccine be used as a primary-series option for adults ages 18 years and older. Novavax, which will be available in the coming weeks, is the first protein subunit COVID-19 vaccine recommended for use in the U.S. Having multiple types of COVID-19 vaccines offers options and flexibility for providers and recipients. The Novavax COVID-19 vaccine’s safety and effectiveness have been confirmed by clinical trials with thousands of individuals in the U.S.

Stay Safe in the Summer Heat
Heat related deaths are preventable. Stay safe and protect your family and our entire community during times of extreme heat this summer.
HEALTH ADVISORY # 199 – CDC Recommends COVID-19 Vaccine for Children 6 Months through 5 Years
On June 18, 2022, the Centers for Disease Control and Prevention (CDC) endorsed the Advisory Committee on Immunization Practices (ACIP) recommendation that children ages 6 months through 5 years should receive COVID-19 vaccination.
HEALTH ADVISORY #198 Amendments to the WV Reportable Disease Rule
The purpose of this advisory is to communicate important changes related to the reporting of communicable diseases in West Virginia. The following summarizes amendments to the West Virginia Legislative Rule, Reportable Diseases, Events and Conditions (64CSR-7)
HEALTH ADVISORY #197 Updated Rabies Preexposure Prophylaxis Recommendations
Human rabies is a preventable viral infection often transmitted following a rabid animal bite. The infection is almost always fatal once symptoms develop, so prevention is critical. There are measures to prevent rabies infection in humans including vaccination of targeted animals, use of personal protective equipment (PPE), avoid aggravation of high-risk mammals, and administration of rabies post-exposure prophylaxis (PEP) following potential rabies exposure.
HEALTH ADVISORY #196 CDC Recommends COVID-19 Booster for Children 5 to 11 Years and Strengthens Recommendations
On May 19, 2021, the Centers for Disease Control and Prevention (CDC) endorsed the Advisory Committee on Immunization Practices (ACIP) recommendation that children 5 to 11 years old should receive a COVID-19 booster five months after their initial Pfizer-BioNTech pediatric vaccine series.
HEALTH ADVISORY #180 CDC Recommends Booster Dose of Pfizer COVID-19 Vaccine for Certain Populations and Those in High-Risk Settings
On September 24, 2021, the Centers for Disease Control and Prevention (CDC) endorsed the Advisory Committee on Immunization Practices (ACIP) recommendation for a booster dose of the Pfizer-BioNTech mRNA COVID-19 vaccine in certain populations and recommended a booster dose for those in high-risk occupational and institutional settings.
HEALTH ADVISORY #178 CDC Recommends Additional Dose of mRNA COVID-19 Vaccine for Immunocompromised People
Studies indicate some immunocompromised people have a reduced immune response following a primary COVID-19 vaccine series compared to vaccine recipients who are not immunocompromised and that a third dose may enhance immune response. For people with moderate to severe immune compromise due to a medical condition or receipt of immunosuppressive medications or treatments, the potential to increase immune response coupled with an acceptable safety profile, support the recommendation for an additional mRNA vaccine dose after an initial 2-dose primary mRNA COVID-19 vaccine series.
HEALTH ADVISORY #171 Point-of-Care Antigen Testing for SARS-CoV-2 in Nursing Homes
This advisory pertains to antigen tests that have been granted a US FDA Emergency Use Authorization (FDA EUA) to detect SARS-CoV-2. The first SARS-CoV-2 antigen tests to receive FDA EUA are authorized for testing symptomatic persons within 5-7 days of symptom onset and there are limited data on antigen test performance in asymptomatic persons. However, given the transmission of SARS-CoV-2 from asymptomatic and pre-symptomatic nursing home residents and healthcare personnel (HCP) with SARS-CoV-2 infection, the WVDHHR is providing considerations for the use of antigen tests for asymptomatic persons during this public health emergency.
HEALTH ALERT #169 New Laboratory Data Reporting Elements for COVID-19 and Death Reporting
The West Virginia Department of Health and Human Resources (DHHR), Bureau for Public Health (BPH) continues to respond to coronavirus disease (COVID-19) in West Virginia. The purpose of this advisory is to update healthcare providers on new national requirements for COVID-19 laboratory test data elements. These added reporting requirements will help assure rapid public health response to COVID-19 across West Virginia communities.
HEALTH ALERT/UPDATE #168 Enpanding Testing for COVID-19: Clinician-Suspected Re-infection
In light of increased testing availability, evolving information on COVID-19 (pre-symptomatic and potentially asymptomatic spread), and increasing community movement and activities, the West Virginia Department of Health and Human Resources (WVDHHR), Bureau for Public Health (BPH) supports expanded provider and community testing for COVID-19. Providers should work with more than a single laboratory to assure ongoing availability of supplies and services. Viral testing supplies should first be obtained through the lab performing the test, but if not available, may be accessed through the West Virginia Office of Laboratory Services (OLS) working through your local health department (LHD).
HEALTH ALERT/UPDATE #166 COVID-19
The West Virginia Department of Health and Human Resources (WVDHHR) and the West Virginia Bureau for Public Health (WVBPH) continue to respond to novel coronavirus disease (COVID-19) in West Virginia. WVBPH is working closely with state agencies, local and federal partners to monitor the ongoing risk of COVID-19 and to ensure the health and protection of state residents. Below is information pertaining to personal protective equipment (PPE) conservation efforts, laboratory and death reporting, online requests for testing through the WV Office of Lab Services, and Pediatric Multisystem Inflammatory Syndrome in Children (MIS-C).
ADVISORY for Self-Isolation of the Kanawha Charleston Health Department
The Governor of West Virginia has ordered a mandatory Stay-at-Home Order, effective on March 24, 2020, at 8:00pm, requiring all citizens to stay at home and limit movements outside their homes beyond essential needs, in order to slow the spread of the novel coronavirus (COVID-19). In a further effort to slow the spread of COVID-19, […]
Guidance for Kanawha Judicial Annex employees
Because of possible COVID exposure of employees in the Kanawha Judicial Annex, the Kanawha-Charleston Health Department recommends employees do the following: Employees must isolate themselves until April 6. Employees should monitor their temperatures twice daily. If fevers increase or they have other symptoms—cough, shortness of breath—they should contact their health care providers. The general definition […]

Coronavirus Disease 2019 (COVID-10) Guidance for Restaurants, Bars and Casinos Closures March 2020
Governor Jim Justice issued an Executive Order, which is now in effect, formally directing all restaurants and bars in West Virginia to limit service to carry out, drive-thru, and delivery operations only. The Executive Order also mandates the closure of all casinos in West Virginia.
Restaurants with carry out, drive-thru, and delivery options will still be able to operate those services, even as their dining rooms are temporarily closed. The order officially went into effect on Wednesday, March 18, 2020, at 12:00 a.m. (midnight).
Continue Reading Here
Health Update # 165 Coronavirus Disease 2019 (COVID-19) Outbreak
Public health authorities in West Virginia continue to monitor the evolving COVID-19 outbreak and engage others in planning forward. Updated assessment/recommendations include the following:
• At this time, West Virginia has no confirmed cases of COVID-19. Community spread is being detected in a growing number of countries, including parts of the U.S. As community spread increases, we anticipate the arrival and local transmission of COVID-19 in our state.
Continue Reading Here
HEALTH ALERT #164 2019 Novel Coronavirus (2019-nCoV) Outbreak
There is a rapidly expanding outbreak in China of respiratory illness caused by a novel (new) coronavirus abbreviated “2019-nCoV”. This virus is spreading from person-to-person in China and exported cases have been detected in a growing number of countries internationally. Imported cases of 2019-nCoV infection in people have been detected in the US. While limited person-to-person spread among close contacts has been detected in the US, there is currently no evidence that 2019-nCoV is actively spreading across US communities.
HEALTH ALERT #163 Update on Vaping-Associated Lung Injuries
• E-cigarette, or vaping, product use associated lung injury (EVALI), previously referred to as vaping-
associated lung injury, should be reported within 24 hours to the local health department (LHD) or to the Bureau for Public Health (BPH) by calling (304) 558-5358 per the Reportable Disease Rule 64-CSR-7.
• Rapid recognition of patients with EVALI, increased understanding of diagnostic and treatment considerations, and timely reporting to public health could reduce morbidity and mortality associated with this injury.
Continue Reading Here
HEALTH ADVISORY #162 Human Immunodeficiency Virus (HIV) Infections Among People Who Inject Drugs — Additional Area Seeing Increase, Others Vulnerable
HIV has long been present across West Virginia, although at low levels. With the state’s opioid and now broader substance use epidemic, West Virginia has been increasingly vulnerable to HIV outbreak(s) among persons who inject drugs (PWID). The sharing of injection drug equipment as well as high-risk sexual practices associated with substance use generate this vulnerability. The West Virginia Bureau for Public Health (BPH) actively monitors HIV diagnoses across the state and works with local and federal partners to prevent, investigate and respond to increases.
Continue Reading Here
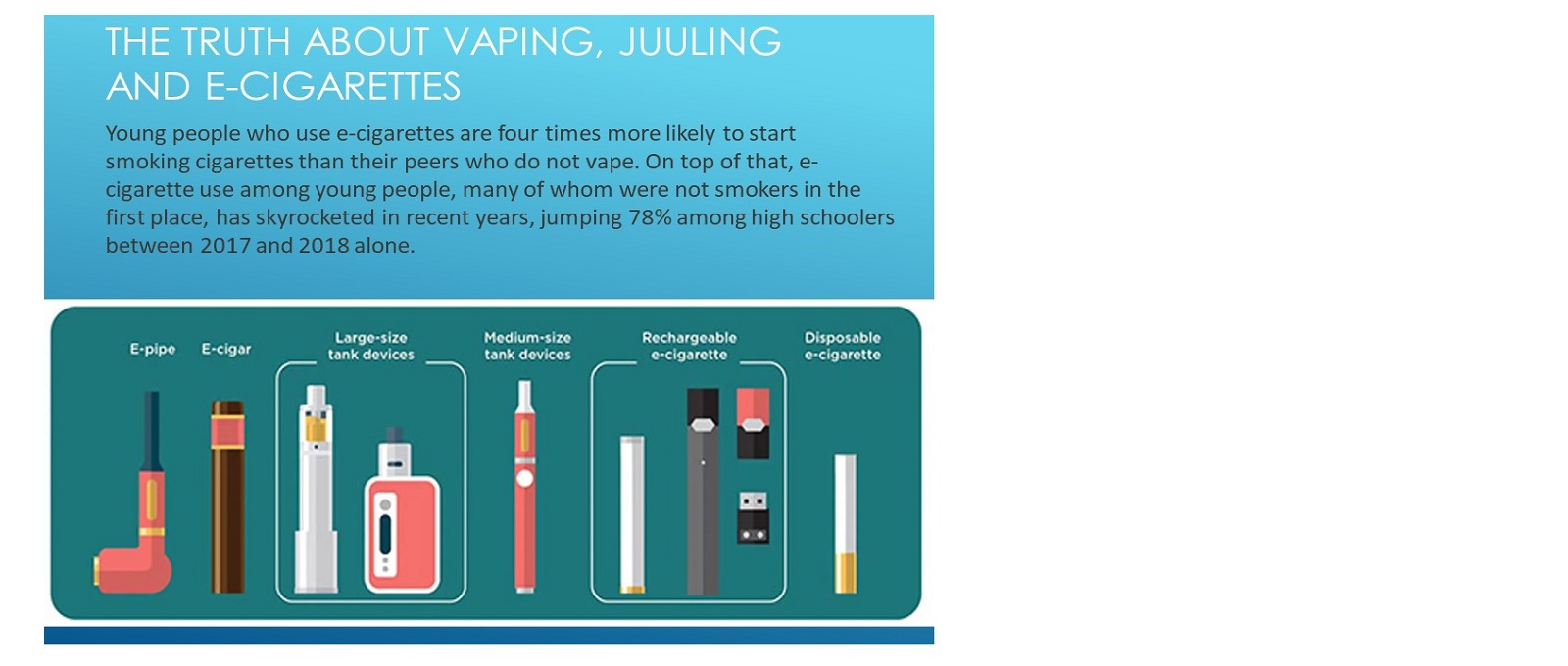
Vaping and e-cigarette information for parents
What’s the bottom line on the risks of e-cigarettes for kids, teens and young adults?
• The use of e-cigarettes is unsafe for kids, teens, and young adults.
HEALTH ALERT #161 Severe Pulmonary Illness Associated with Vaping
Multiple states have reported cases of severe pulmonary illness primarily among adolescents and young adults who report vaping (use of e-cigarette and similar devices to inhale aerosolized liquid). Patients reported vaping both nicotine and tetrahydrocannabinol (THC)-containing products. Product names are unknown. The Bureau for Public Health has begun to investigate sporadic cases. During 2017-2018, current e-cigarette use in the
U.S. increased by 78% (from 11.7% to 20.8%) among middle and high school students. It is important to note that West Virginia has a high rate of youth vaping. The Bureau for Public Health is calling for expanding surveillance to more readily identify any illnesses and medical incidents caused by vaping.
Continue Reading Here
HEALTH ADVISORY #160 24th World Scout Jamboree
This Health Advisory serves as a reminder to all healthcare providers and laboratorians to report outbreaks, communicable diseases, and unusual conditions or emerging infectious diseases during the 24th World Scout Jamboree (WSJ) as required by the West Virginia Legislative Rule for Reportable Diseases, Events and Conditions (64CSR7). West Virginia will host the WSJ July 22, 2019 – August 2, 2019 at the Summit Bechtel Family National Scout Reserve (SBR) in Mount Hope, West Virginia. The WSJ is anticipated to be one of the largest mass gatherings in the State’s history with over 43,000 scouts from 138 countries. Additionally, SBR will be open to day visitors, increasing the number of people on the reserve to approximately 50,000. The event is expected to bring an increased number of visitors to Fayette, Raleigh and surrounding counties.
HEALTH ADVISORY #159 Vector-borne Disease Season
With the spring season comes an increase in mosquito and tick activity in West Virginia. Vector-borne diseases occur annually beginning in early spring and peaking in August and September when people and arthropod vectors are most active outdoors. Health providers can educate patients on preventive measures, including use of recommended insect repellents with DEET and permethrin, to prevent bites and control diseases spread by mosquitoes and ticks.
HEALTH ADVISORY #158 Recommendations for Routine HIV Testing and Available Resources for Healthcare Providers
The Bureau for Public Health, in collaboration with local and federal partners, is responding to an increase in newly diagnosed cases of HIV among persons who inject drugs (PWID) in West Virginia. Public health partnerships, expanded HIV testing, early diagnosis and linkage to care, harm reduction services, and initiation of or referral to substance use disorder recovery as applicable are all critical to preventing and addressing HIV in the community. Thanks to new and better treatments, people with HIV who remain in care are now living longer – and with a better quality of life – than ever before.
HEALTH ADVISORY #157 Measles
The Centers for Disease Control and Prevention (CDC) has identified an increased number of measles cases and outbreaks in the United States since January 1, 2019. The last case of measles reported in West Virginia was in 2009 following international travel. Clinicians are encouraged to continue vigilance for cases of measles among all age groups and to report suspected cases of measles immediately to their local health department (LHD) as per the West Virginia Reportable Disease Rule (64 CSR 7). Additionally, clinicians should take this opportunity to ensure patients are up-to-date on measles vaccination (MMR). MMR information can be found at https://www.cdc.gov/vaccines/vpd/measles/index.html.
Health Advisory
NEW recommendations for ALL health providers on HIV screening, testing, and follow-up care.
HEALTH ALERT #156 Acute Flaccid Myelitis
West Virginia has confirmed a case of Acute Flaccid Myelitis (AFM). AFM is a rare but serious condition that affects the nervous system, causing muscles and reflexes in the body to become weak. The last confirmed case of AFM in West Virginia was in 2016. The Centers for Disease Control and Prevention (CDC) recognized AFM in 2014 as a syndrome following increased reports of acute limb weakness and began surveillance for this condition. Clinicians are encouraged to continue vigilance for cases of AFM among all age groups and to immediately report suspected cases of AFM to their local health department (LHD) as per the West Virginia Reportable Disease Rule (64 CSR 7).
HEALTH ADVISORY #155 Increase in New HIV infections Among Persons Who Inject Drugs
The West Virginia Bureau for Public Health is investigating an increase in newly diagnosed human immunodeficiency virus (HIV) cases in the state among persons who inject drugs (PWID). Historically, male-to-male sexual contact has been the predominant reported risk factor for becoming infected with HIV. Since 2018, we have seen an increase in the number of newly diagnosed cases of HIV with injection drug use (IDU) reported as a risk factor statewide.
Continue Reading Here
Update—Outbreak of Life-threatening Coagulopathy Associated with Synthetic Cannabinoids Use
The Centers for Disease Control and Prevention (CDC) is providing information on: 1) the current status of a multistate outbreak of coagulopathy from exposure to synthetic cannabinoid products containing a vitamin K-epoxide cycle antagonist, brodifacoum; 2) the emergence of 2 new clinical scenarios; and 3) recommendations to help clinicians make decisions related to these 2 new clinical scenarios.
This is an update to the Health Alert Network (HAN) advisory released on May 25, 2018 titled Outbreak of Life-threatening Coagulopathy Associated with Synthetic Cannabinoids Use (https://emergency.cdc.gov/han/han00410.asp).
HEALTH ADVISORY #153 Update on Supply of Hepatitis A Vaccine, Prioritization for State- Supplied Vaccine, and Clinical Considerations
The supply of adult hepatitis A vaccine continues to be constrained nationally due to the hepatitis A outbreak among adults in several states resulting in substantially increased demand for adult hepatitis A vaccine. There are, however, adequate supplies of the pediatric formulation of hepatitis A vaccine, so routine vaccination of all children is strongly encouraged to prevent transmission of hepatitis A virus (HAV) to children.
HEALTH ADVISORY #152 Cluster of Legionnaire’s Disease in Northern Panhandle
Since late September 2018, six (6) cases of confirmed Legionella pneumonia (also known as Legionnaire’s Disease) have been reported among persons that work at a facility in the Northern Panhandle. These cases are tightly clustered in time and there may be potentially associated cases that have not yet been identified. To date, the common occupational exposure remains the only epidemiologic link between cases. Federal, state and local public health officials are closely working to identify potential sources of exposure and to mitigate risk of additional cases.
HEALTH ADVISORY #151 Influenza Season 2018-2019
September 30, 2018 officially began the 2018-2019 influenza season. Since the 2017-2018 influenza season was one of the most severe seen in several years, it is important to be prepared for the upcoming season. The West Virginia Bureau for Public Health would like to remind stakeholders of the mandatory reporting requirements for the 2018-2019 season.
Continue Reading Here
HEALTH ADVISORY #150 Vigilance for Cases of Acute Flaccid Myelitis
Since acute flaccid myelitis (AFM) was first recognized in the United States in 2014, only two confirmed cases have been reported in West Virginia (both in 2016). Though no cases have been reported since then, healthcare providers are encouraged to continue vigilance for cases of AFM among all age groups and report suspected cases of AFM to their local health department (LHD) as per the WV Reportable Disease Rule (64 CSR 7).
AFM is characterized by a sudden onset of weakness in one or more limbs following a respiratory or febrile illness. Magnetic resonance imaging (MRI) reveals distinct abnormalities of the spinal cord gray matter.
Advice to Clinicians about Leptospirosis in U.S. Travelers Returning from Northern Israel
The Israeli Ministry of Health is reporting an outbreak of leptospirosis in persons with exposure to natural water sources in the Golan Heights region of northern Israel after July 1, 2018. As of September 6, 2018, three persons with leptospirosis who traveled to Israel have been identified in the United States, with additional suspected cases reported and under investigation. Early symptoms of leptospirosis include fever, headache, chills, muscle aches, vomiting, diarrhea, cough, conjunctival suffusion (conjunctival redness without exudates), jaundice, and sometimes a rash. Clinicians should consider leptospirosis as a diagnosis in any patient who develops an acute febrile illness within 4 weeks of travel to one of the areas in northern Israel listed below since July 1, 2018.
KCHD recommends Hep A vaccinations for Lee Street Lounge cookout patrons
Patrons of the Lee Street Lounge, located at 1111 Lee St., E., in Charleston, who ate food and had beverages during a Monday, Aug. 6, cookout at the facility may have been exposed to hepatitis A and should be vaccinated by Aug. 20. Vaccinations received within two weeks of exposure can prevent the liver disease. Those who participated in cookouts held July 23 and July 30 should monitor for symptoms. An individual associated with preparing the food has been diagnosed with Hepatitis A
HEALTH ADVISORY #149 Information for Healthcare Providers and Emergency Departments on the Hepatitis A Outbreak
The West Virginia Bureau for Public Health (BPH) continues to investigate cases of hepatitis A as part of the multi-state outbreak that is occurring nationally. Persons who use injection and non-injection drugs, persons with unstable housing or are homeless, persons with recent history of incarceration, and men who have sex with men (MSM) are at considerable risk for infection. There is continued transmission of hepatitis A in West Virginia due to increasing case counts and the long incubation period of the disease.
Suggestions offered for dealing with mosquito-borne illnesses
As summer takes hold, residents are becoming more concerned about the annual mosquito-borne illness problem. According to Christina Hinkle, RN, Kanawha-Charleston Health Department’s director of clinic services, “West Nile virus is a mosquito-borne illness that causes no symptoms in approximately 80 percent of the people who are infected.”
As many as 20 percent of those infected with West Nile virus will have symptoms including body aches, fever, headaches, nausea, vomiting and sometimes swollen lymph glands or skin rashes on the stomach or back. About 1 in 150 people develop more severe illnesses that may cause neurological impairment.
HEALTH ADVISORY #148 Voluntary NonOpioid Advanced Directive
The purpose of this Health Advisory is to provide guidance regarding the Voluntary NonOpioid Advanced Directive (VNOAD) form as established in Senate Bill 273, the Opioid Reduction Act of 2017 (“Act”), specifically W.Va. Code §16-54-2.
HEALTH ADVISORY #147 Update on the Multi-State Hepatitis A Outbreak
As of June 8, 2018, West Virginia has identified 203 cases from 11 counties. Most cases have been reported from Cabell, Kanawha, and Putnam counties. Hospitalization remains high at about 70%. BPH is working closely with local health departments to obtain the most complete epidemiologic data available on cases to prevent secondary cases among close contacts. Vaccination of high-risk groups continues to be an important strategy in preventing new cases. In early June, BPH collaborated with the Regional Jail Authority to vaccinate inmates at the Western and Southcentral Regional Jails
KCHD offers warm weather guidance
“Now is the time to prepare for hot temperatures that bring illness and even death every year,” according to Christina Hinkle, director of clinic services, at the Kanawha-Charleston Health Department and the Putnam County Health Department.
“Take measures to stay cool. Remain hydrated and keep informed. Getting too hot can make you sick. You can become ill from the heat if your body can’t compensate for it and properly cool you off,” Hinkle said.
HEALTH ADVISORY #146 Vectorborne Disease Season
With the spring season comes an increase in mosquito and tick activity in West Virginia. Vectorborne disease season typically runs from May to November each year. Human cases of vectorborne disease are most frequently reported during summer since people are most active and outdoors when the weather is warm.
Outbreak of Life-threatening Coagulopathy Associated with Synthetic Cannabinoids Use
The Centers for Disease Control and Prevention (CDC) is providing information on: 1) the current status of a multistate outbreak of coagulopathy from exposure to synthetic cannabinoid products containing a vitamin K-dependent antagonist agent, such as brodifacoum; 2) signs and symptoms of presenting patients from this outbreak and which patients are at risk; 3) laboratory testing options that are available to help identify and classify cases; 4) available resources that may help clinicians make decisions; and 5) to whom to report possible cases.
HEALTH ADVISORY #145 Increased Cases of Hepatitis A Among Persons Who Are Homeless and Persons Who Use Illicit Drugs
West Virginia is reporting an increase in hepatitis A virus (HAV) cases in some counties, namely Kanawha and Putnam. Several have been molecularly linked to the multi-state outbreak in which genotype 1B is the HAV strain of concern; other cases have been epidemiologically-linked to cases from Kentucky.
HEALTH ADVISORY #144 Enhanced Surveillance for Hepatitis A Cases Among Homeless and Persons Who Inject Drugs
Multiple state (California, Kentucky, Michigan, and Utah) and local health departments have been reporting hepatitis A virus (HAV) (genotype 1B) outbreaks among homeless persons, persons who use injection and non-injection drugs, and their close contacts since 2016. At this time, the West Virginia Bureau for Public Health (BPH) has not seen an increase in reported HAV cases.
HAV is a vaccine-preventable disease that is usually transmitted person-to-person through the fecal-oral route or consumption of contaminated food or water. People who are homeless are at particularly high-risk due to lack of access to clean toilets and handwashing facilities. HAV does not result in chronic infection and can range from a mild illness lasting a few weeks to severe illness lasting several months. Symptoms include acute onset of fever, nausea, vomiting, abdominal pain, diarrhea, dark urine, clay-colored stool, and jaundice
HEALTH ADVISORY #142 Vigilance for Cases of Acute Flaccid Myelitis
Since acute flaccid myelitis (AFM) was first recognized in the United States in 2014, only two confirmed cases have been reported in West Virginia (both in 2016). Though no cases have been reported in 2017, healthcare providers are encouraged to continue vigilance for cases of AFM among all age groups and report suspected cases of AFM to their local health departments. Reporting of cases will help public health monitor the occurrence of AFM to better understand factors associated with this illness.
HEALTH ADVISORY #143 Widespread Influenza Activity and Severity in West Virginia
Influenza activity has increased significantly in the United States in recent weeks. West Virginia is characterized as having widespread activity. This influenza season is notable for the sheer volume of ill cases that has caused stress to health systems. The Centers for Disease Control and Prevention (CDC) released a Health Advisory (https://emergency.cdc.gov/han/han00409.asp) that warned of a high-severity influenza A (H3N2) season and resulting clinical implications. As of January 6, 2018, 20 pediatric influenza deaths were reported nationally, none in West Virginia.
HEALTH ADVISORY #141 Dramatic Increase in Lyme Disease Cases
West Virginia will report the highest number of Lyme disease cases on record in 2017. As of November 8, 2017, 571 confirmed and probable Lyme disease cases have been reported compared to the previous record of 368 cases in 2016. The geographic distribution of cases has also increased. As of November 8, 2017, 45 counties have reported at least one confirmed or probable Lyme disease case, up from 11 counties in 2012. This increase could be the result of increased reporting, increase in the number of Ixodes scapularis ticks infected with Borrelia burgdorferi in West Virginia, or a recent change in the national surveillance case definition for Lyme disease. Based on the change, West Virginia is considered a high incidence Lyme disease state, and the presence of an erythema migrans (EM) with known exposure in the state is sufficient for a patient to be classified as a confirmed surveillance case.
Advice for Health Care Providers Treating Patients in or Recently Returned from Hurricane-Affected Areas
The Centers for Disease Control and Prevention (CDC) is working with federal, state, territorial, and local agencies and global health partners in response to recent hurricanes. CDC is aware of media reports and anecdotal accounts of various infectious diseases in hurricane-affected areas, including Puerto Rico and the US Virgin Islands (USVI). Because of compromised drinking water and decreased access to safe water, food, and shelter, the conditions for outbreaks of infectious diseases exist.
Rifampin/Penicillin-Resistant Strain of RB51 Brucella Contracted from Consumption of Raw Milk
The Texas Department of State Health Services, with assistance from CDC, is investigating Brucella RB51 exposures and illnesses that may be connected to the purchase and consumption of raw (unpasteurized) milk from K-Bar Dairy in Paradise, Texas. Symptoms of brucellosis can include: fever, sweats, malaise, anorexia, headache, fatigue, muscle & joint pain, and potentially more serious complications (e.g., swelling of heart, liver, or spleen, neurologic symptoms).
Hurricane Harvey – Clinical Guidance for Carbon Monoxide (CO) Poisoning
Carbon monoxide (CO) is an odorless, colorless, poisonous gas that can cause sudden illness and death if present in sufficient concentration in the ambient air. During a significant power outage, persons using alternative fuel or power sources such as generators or gasoline powered engine tools such as pressure washers might be exposed to toxic CO levels if the fuel or power sources are placed inside or too close to the exterior of the building causing CO to build up in the structure. The purpose of this HAN advisory is to remind clinicians evaluating persons affected by the storm to maintain a high index of suspicion for CO poisoning. Clinicians are advised to consider CO exposure and take steps to discontinue exposure to CO. Clinicians are also advised to ask a patient with CO poisoning about other people who may be exposed to the same CO exposure, such as persons living with or visiting them so they may be treated for possible CO poisoning.
Increase in West Nile Virus Activity in Mosquitoes
Based on data collected from May 24, 2017 to July 26, 2017, the West Virginia Department of Health and Human Resources, Bureau for Public Health Mosquito Surveillance Program is reporting several West Nile virus (WNV) positive mosquito pools across West Virginia with the greatest number of infected mosquitoes from Cabell County. WNV positive mosquito pools have also been detected in Berkeley, Fayette, Kanawha, Putnam, Wayne, Wetzel, and Wood counties this season.
Increase in Reported cases of Cyclospora Cayetanensis Infection, United States, Summer 2017
The Centers for Disease Control and Prevention (CDC), State and Local Health Departments, and the Food and Drug Administration (FDA) are investigating an increase in reported cases of cyclosporiasis. The purpose of this HAN Advisory is to notify public health departments and healthcare facilities and to provide guidance to healthcare providers of the increase in reported cases. Please disseminate this information to healthcare providers in hospitals and emergency rooms, to primary care providers, and to microbiology laboratories.
Increase in New HIV Infections Reported in West Virginia
The Bureau for Public Health is investigating an increase in newly diagnosed human immunodeficiency virus (HIV) cases in the State among persons at high risk for infection. In addition to syphilis (reported among several of the cases), this at-risk population remains at high risk for hepatitis B and C infection.
2017 National Boy Scout Jamboree
This Health Advisory serves as a reminder to all healthcare providers and laboratorians to report outbreaks, communicable diseases, and unusual conditions or emerging infectious diseases during the National Boy Scout Jamboree as required by the West Virginia Legislative Rule for Reportable Diseases, Events and Conditions (64CSR7). West Virginia will host the 2017 National Boy Scout Jamboree from July 19-28, 2017 at the Summit Bechtel Family National Scout Reserve in Mount Hope, West Virginia
HEALTH ADVISORY #137 Legionnaire’s Disease in West Virginia
During the months of May and June 2017, West Virginia experienced more cases than expected of Legionnaire’s Disease (Legionella pneumonia).
Patients Receiving Eculizumab (Soliris®) at High Risk for Invasive Meningococcal Disease Despite Vaccination
Summary
Eculizumab (Soliris®) recipients have a 1,000 to 2,000-fold greater risk of invasive meningococcal disease compared to the general U.S. population. The Food and Drug Administration (FDA)-approved prescribing information for eculizumab includes a black box warning for increased risk of meningococcal disease, and the Advisory Committee on Immunization Practices (ACIP) recommends meningococcal vaccination for all patients receiving eculizumab. Recent data show that some patients receiving eculizumab who were vaccinated with the recommended meningococcal vaccines still developed meningococcal disease, most often from nongroupable Neisseria meningitidis, which rarely causes invasive disease in healthy individuals.
Potential for Falsely Low Blood Lead Test Results from LeadCare Analyzers
Children are particularly vulnerable to lead exposure due to the effect on their developing brains and organ systems. The U.S. Food and Drug Administration (FDA) and Centers for Disease Control and Prevention (CDC) are warning Americans that certain lead tests manufactured by Magellan Diagnostics may provide inaccurate results for some children and adults in the United States. FDA is now warning that M age llan Dia gn ostics’ Le adC are® ana l yzer s (LeadCare, LeadCare II, LeadCare Ultra and LeadCare Plus) should no longer be used with venous blood samples due to the potential for falsely low test results.
Potential for Falsely Low Blood Lead Test Results from LeadCare® Analyzers
The U.S. Food and Drug Administration (FDA) has issued a safety communication warning about the use of Magellan Diagnostics’ LeadCare® analyzers (LeadCare, LeadCare II, LeadCare Ultra and LeadCare Plus) with venous blood samples because they might result in falsely low test results. FDA is now advising that Magellan Diagnostics’ LeadCare® analyzers should no longer be used with venous blood samples. The safety alert does not apply to capillary blood lead test results collected by fingerstick or heelstick. The purpose of this Health Advisory is to notify state and local health departments, healthcare providers, and laboratories about CDC’s re-testing guidance in light of the safety alert.
Mosquito-Borne Disease Season
Mosquito-borne diseases occur annually in West Virginia beginning in early spring and peaking in August and September, coinciding with mosquito activity. Arboviral infections, particularly La Crosse encephalitis (LAC) and West Nile virus (WNV), are endemic mosquito-borne diseases identified in West Virginia. In 2016, eight LAC cases and one WNV case were reported in West Virginia. Persons with severe arboviral infections will often have symptoms of encephalitis. Please be vigilant in identifying such cases, and ensure that all hospitalized patients with encephalitis undergo appropriate arboviral disease testing during mosquito season.
Prolonged IgM Antibody Response in People Infected with Zika Virus: Implications for Interpreting Serologic Testing Results for Pregnant Women
In July 2016, CDC issued Interim Guidance for Health Care Providers Caring for Pregnant Women with Possible Zika Virus Exposure – United States, July 2016 (https://www.cdc.gov/mmwr/volumes/65/wr/mm6529e1.htm) that includes Zika virus immunoglobulin M (IgM) testing of pregnant women. However, some flavivirus infections can result in prolonged IgM responses (>12 weeks) that make it difficult to determine the timing of infection, especially in testing of asymptomatic people. Emerging epidemiologic and laboratory data indicate that Zika virus IgM can persist beyond 12 weeks in a subset of infected people. Therefore, detection of IgM may not always indicate a recent infection. Although IgM persistence could affect IgM test interpretation for all infected people, it would have the greatest effect on clinical management of pregnant women with a history of living in or traveling to areas with Zika virus transmission. Pregnant women who test positive for IgM antibody may have been infected with Zika virus and developed an IgM response before conception.
Tickborne Disease Season
Tickborne diseases occur annually in West Virginia with most cases developing symptoms between April and September. Lyme disease is the most commonly reported tickborne disease in West Virginia. In 2016, West Virginia recorded 368 Lyme disease cases, the most ever in a single year. Counties in the northwestern and southwestern parts of the state have had increasing case counts in recent years. In 2016, 43 counties reported at least one confirmed or probable Lyme disease case. Based on new national reporting standards, West Virginia is considered a high incidence Lyme disease state.
CDC Recommendations for Diagnosing and Managing Shigella Strains with Possible Reduced Susceptibility to Ciprofloxacin
This Health Advisory describes the identification of emerging Shigella strains with elevated minimum inhibitory concentration values for ciprofloxacin and outlines new recommendations for clinical diagnosis, management, and reporting, as well as new recommendations for laboratories and public health officials. Current interpretive criteria provided by the Clinical and Laboratory Standards Institute (CLSI) categorize these strains as susceptible to ciprofloxacin, which is a fluoroquinolone antibiotic and a key agent in the management of Shigella infections.
Increased Screening Recommendations for HIV Outbreak Detection Among Persons Who Inject Drugs
The epidemic of non-prescription opioid addiction has led to an increase in injection drug use, hepatitis C virus (HCV) infections, and hepatitis B virus (HBV) infections in West Virginia. Though West Virginia is a low incidence state for human immunodeficiency virus (HIV) infections, several counties have been deemed at high-risk for an HIV outbreak similar to the one in Scott County, Indiana in 2015 predominantly among people who inject drugs (PWID). In order to detect an outbreak of HIV among PWID, increased screening is necessary.


